 (Source: Oleg Lavrentovich, Liquid Crystal Institute, Kent State University)
(Source: Oleg Lavrentovich, Liquid Crystal Institute, Kent State University)
The Big Picture
When you go shopping for a flat-panel television these days, the vast majority of available models are LCD TVs. Today’s LCDs are super-slim, super-bright, and super-colorful thanks to many improvements made since Sharp introduced the first commercial LCD TV—with a screen measuring a whopping 14 inches—in 1988.
But exactly how does an LCD TV work? What improvements have been made over the last quarter century that led to this technology’s overwhelming dominance in the flat-panel market? Let’s begin with a broad overview…
Like all video displays these days, LCD TVs form an image in an array of tiny pixels. In most cases, the array is 1920 pixels horizontally by 1080 pixels vertically, though lower-end sets are sometimes 1280×720 or 1366×768. The pixels in an LCD TV are not emissive—that is, they do not emit light on their own as they do in plasma and OLED TVs. Instead, they allow more or less light to pass through them. As a result, LCD TVs are sometimes called transmissive displays because they transmit light through the pixels.
The light in an LCD TV comes from a backlight at the back of the screen. In the past, this backlight consisted of small fluorescent tubes called CCFLs (cold-cathode fluorescent lights), which are much like small versions of the fluorescent lights in a typical office. However, in modern LCD TVs, this type of backlight has been mostly replaced by LEDs (light-emitting diodes), because they have no mercury content, use electricity more efficiently, and can be controlled to a greater degree than CCFLs. In most cases, the LEDs are placed at the edges of the screen in order to make the entire TV thinner, though a few models have an array of LEDs directly behind the screen.
By the way, an LCD TV with LED backlighting is often called an “LED TV” by its manufacturer, a confusing marketing misnomer that leads consumers to think it’s a different display technology than LCD. It’s not—an LED TV is simply an LCD TV with an LED backlight, which is why I normally refer to them as “LED/LCD TVs.”
No matter what type of backlight is used—CCFLs, LEDs on the edges, or LEDs directly behind the screen—the light is diffused by a special layer of plastic to make it as uniform as possible across the entire area of the screen. The light then passes through the pixels, each of which can be independently controlled to pass more or less light through it as well as filter the light into any desired color.
 Fig. 1: Layers of an LCD TV (Source: Sony Electronics)
Fig. 1: Layers of an LCD TV (Source: Sony Electronics)
In Figure 1, from Sony, you can see the various layers of an LCD TV using HCFL (hot-cathode fluorescent light, similar in principle to CCFL) and LED backlights behind the screen and on the edges. In this example, Sony uses red, green, and blue LEDs behind the screen, but virtually all such backlights use white LEDs these days.
The TV’s electronics control the brightness and color of each pixel and process the incoming video signal. For example, if the number of pixels in the signal does not match the number of pixels on the screen, the processor scales the image, adding or subtracting pixels to match the resolution of the screen. If the signal is interlaced—that is, only half of each video frame is sent at a time—the processor deinterlaces it so the TV can display an entire video frame.
How Can a Crystal be Liquid?
The very name of this technology is mysterious. Liquid crystal? What the heck is that? It’s a type of material that exhibits characteristics of both a liquid and a crystal. This material was first discovered in the late 1880s, but it wasn’t until the mid-1960s that liquid crystal was discovered to change its optical properties—that is, how light passes through it—according to an applied electric field. This is the feature that allows liquid crystal to be used in visual displays of many types, including televisions—hence the term “liquid crystal display” or LCD.
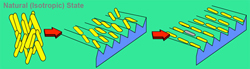 Fig. 2: Nematic liquid-crystal molecules (Source: Sharp Electronics)
Fig. 2: Nematic liquid-crystal molecules (Source: Sharp Electronics)
Basically, liquid-crystal materials consist of rod-shaped polymer molecules that naturally form thin layers, unlike normal liquids in which the molecules are distributed much more randomly. And the molecules in certain types of this material, called nematic liquid crystal, align themselves in roughly the same orientation from one layer to the next, as shown on the left in Figure 2 above. Even more interestingly, if nematic liquid-crystal material is placed against a solid surface etched with microscopic grooves, the molecules next to the surface align themselves with the grooves, as shown in the center and on the right of Figure 2, and the molecules in the layers above align themselves with the ones below, forming a crystal-like lattice.
The actual display panel in an LCD TV—commonly called “the glass”—consists of several layers (not to be confused with the molecular layers in the liquid-crystal material itself). At the core of the panel is a thin film of nematic liquid crystal (a total of less than 5cc in a 42-inch TV) sandwiched between two transparent plates called alignment layers or directors, which are etched with microscopic, parallel grooves.
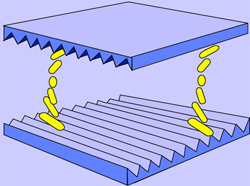 Fig. 3: Nematic liquid-crystal molecules twisting between alignment layers (Source: Sharp Electronics)
Fig. 3: Nematic liquid-crystal molecules twisting between alignment layers (Source: Sharp Electronics)
As illustrated in Figure 3, on one director plate, the grooves are horizontal, while on the other plate, they are vertical. As a result, the nematic liquid-crystal molecules next to the plate with horizontal grooves align themselves horizontally, while the molecules next to the plate with vertical grooves align themselves vertically. The molecules between them are forced to form a spiral as they try to align themselves with their neighbors. As a result, this configuration is called “twisted nematic.”
The director/liquid-crystal/director assembly is further sandwiched between two more plates that polarize light passing through them. Before I explain why this is important and how it relates to the spiral formation of the liquid-crystal molecules, I need to make sure you understand the concept of polarization.
Polarization Explained
As you may know, light is a form of electromagnetic radiation that travels as a wave, somewhat akin to water waves. Unlike water waves, however, the crests and troughs of a light wave can be horizontal, vertical, or any angle in between. The angle formed by a light wave’s crests and troughs with respect to horizontal and vertical is called its polarization.
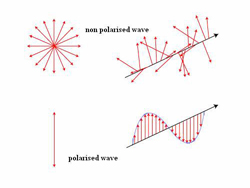 Fig. 4: The polarization of most light is random, as shown in the upper part of this diagram. (Source: esemenyhorizont.uw.hu)
Fig. 4: The polarization of most light is random, as shown in the upper part of this diagram. (Source: esemenyhorizont.uw.hu)
It’s also important to know that the light from most sources does not have a single polarization. Instead, each photon—a “particle” of light—can have a different, random polarization, as shown in the upper portion of Figure 4.
A polarizer allows only light waves with a certain polarization—that is, waves with crests and troughs at a certain angle—to pass through them. For our purposes, a polarizer can be either horizontal or vertical, so only light with horizontal or vertical polarization gets through. Light waves with different polarizations are at least partially blocked; only the horizontal or vertical portion of each photon passes through the polarizer, depending on its orientation.
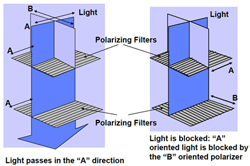 Fig. 5: Parallel and perpendicular polarizers (Source: Sharp Electronics)
Fig. 5: Parallel and perpendicular polarizers (Source: Sharp Electronics)
Normally, if light passes through a horizontal polarizer, it is then completely blocked by a subsequent vertical polarizer, as shown on the right in Figure 5. If you stacked a horizontal and vertical polarizer together and looked at a light source through them, you’d see only black, since no light would pass through both polarizers.
Most of us are familiar with polarized sunglasses, which allow vertically polarized light to pass while blocking horizontally polarized reflections, which cuts glare reflected from horizontal surfaces like the road and dashboard of the car. If you place the lenses of two such sunglasses next to each other and rotate one of them 90 degrees, you see virtually no light pass through both lenses.
Polarize This!
Okay, let’s get back to the two polarizer plates in LCD TVs. One of them is a horizontal polarizer, while the other one is a vertical polarizer. It would seem that all the light from the backlight would be blocked from passing through the entire structure, but it isn’t. Why not?
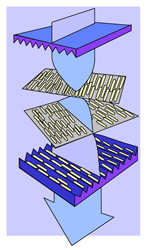 Fig. 6: Light’s polarization follows the liquid-crystal molecules’ spiral. (Source: Sharp Electronics)
Fig. 6: Light’s polarization follows the liquid-crystal molecules’ spiral. (Source: Sharp Electronics)
Because a light wave’s polarization is affected by the orientation of liquid-crystal molecules. As shown in Figure 6, as the polarized light passes through the liquid crystal, the polarization follows the molecules’ spiral orientation, and it ends up in the opposite polarization by the time it gets to the second plate, and so passes through that plate. In other words, light passes through the polarizer/liquid-crystal/polarizer sandwich, which it wouldn’t if the liquid crystal wasn’t there.
Now, recall that liquid crystal changes its optical properties when it’s exposed to an electric field. Specifically, the molecules align themselves with the electric field instead of the grooves in the plates. If the field is weak, the molecular realignment is slight, but as the field gets stronger, the realignment becomes more pronounced.
In the absence of an electric field, imagine that the liquid-crystal molecules form little ladders that have been twisted, forming a double helix much like DNA. When an electric field is applied, the rungs in the middle of the ladders become more vertical, while the rungs next to the alignment plates remain fixed in the grooves. The stronger the electric field, the more the molecular rungs move into a vertical orientation.
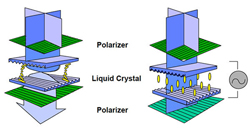 Fig. 7: Without an electric field, light passes through; with an electric field, the molecules reorient, blocking the light. (Source: Sharp Electronics)
Fig. 7: Without an electric field, light passes through; with an electric field, the molecules reorient, blocking the light. (Source: Sharp Electronics)
The more vertical the molecules are, the less the incoming light changes its polarization, which means that less light passes through the second polarizer. With no electric field, all the light passes through, as shown on the left in Figure 7; with a strong field, no light gets through, as shown on the right. This allows the liquid crystal to act like a shutter, letting more or less light pass through the entire sandwich depending on the strength of the electric field.
For all this to work, there must be a light source behind the polarizer/liquid-crystal/polarizer sandwich—the backlight. I’ll get into the different types of backlights more deeply in the next installment. For now, all you need to know is that a large, flat, (hopefully) uniform light source sits behind the polarizer/liquid-crystal/polarizer sandwich, and more or less light from that source passes through the sandwich according to the strength of the electric field applied to it.
Sub-Pixel Magic
Of course, a large flat panel that gets uniformly brighter and darker is hardly useful as a video display. So the liquid-crystal material is actually separated into tiny cells that can be controlled individually. Each cell is called a sub-pixel, and in most LCD TVs, three sub-pixels form one full-color pixel. In a TV with 1920×1080 resolution, there are 2,073,600 pixels, or 6,220,800 sub-pixels.
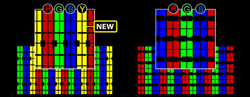 Fig. 8: Sharp adds a yellow sub-pixel in its Quattron LCD TVs. (Source: Sharp Electronics)
Fig. 8: Sharp adds a yellow sub-pixel in its Quattron LCD TVs. (Source: Sharp Electronics)
Each sub-pixel is either red, green, or blue, and each full pixel includes one of each sub-pixel, as shown on the right in Figure 8. (Sharp adds a fourth, yellow sub-pixel in its Quattron LCD TVs, as shown on the left in the diagram above, for a total of 8,294,400 sub-pixels in a 1080p set.) If the light from the backlight is white—which it is—how do the sub-pixels acquire their color? Each sub-pixel cell includes a tiny color filter, either red, green, or blue (or yellow in Quattron sets), located after the light passes through the second polarizer.
The sub-pixels can be arranged in various ways, such as a delta or stripe configuration as shown in Figure 9. The delta configuration is better for video, while the stripe arrangement is better for text. However, the stripe layout is easier and cheaper to manufacture, and the pixels of modern TVs are so small that the sub-pixel arrangement doesn’t make a lot of difference in visual performance. Thus, the vast majority of LCD TVs today use the stripe configuration.
Now, you can probably see how a full-color image is formed on the screen. The amount of light from the backlight that passes through each sub-pixel is independently controlled by applying an electric field of varying strength, changing the mix of red, green, and blue that makes up each pixel.
For example, if no field is applied to any of the sub-pixels, the light passes through all of them at maximum intensity, and the result is a bright white pixel as all three colors combine equally. If a strong field is applied to all three sub-pixels, very little light passes through any of them, resulting in a black pixel. Changing the field applied to each sub-pixel independently lets more or less light through each one, controlling the corresponding pixel’s color and brightness.
Thin-Film Transistors
So how is the electric field generated and controlled for each sub-pixel? In the early days of LCD displays, each alignment layer included a series of long, parallel electrodes—horizontal on one plate and vertical on the other plate—with one electrode for each row and column of sub-pixels, as shown in Figure 10. When a voltage was applied to one row and one column, a field was generated at the intersection point.
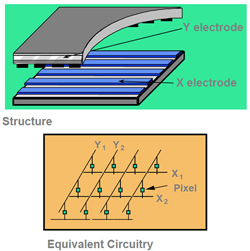 Fig. 10: Passive matrix (Source: Sharp Electronics)
Fig. 10: Passive matrix (Source: Sharp Electronics)
Unfortunately, this electrode structure, called a passive-matrix design, is not sufficient for a high-quality video display, because the field leaks into the surrounding sub-pixels, causing blurring. Also, the voltage needed to change the orientation of the liquid-crystal molecules is very low, causing the molecules to change their orientation too slowly, which resulted in ghosting.
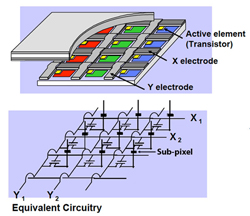 Fig. 11: Active matrix (Source: Sharp Electronics)
Fig. 11: Active matrix (Source: Sharp Electronics)
To solve these problems, a thin-film transistor (TFT) is deposited within each sub-pixel on the first alignment plate (the one closer to the backlight). Also, both sets of addressing electrodes are deposited on the same plate, with the horizontal electrodes insulated from the vertical ones, as shown in Figure 11. This arrangement, called an active-matrix design, limits the electric field to a much smaller area, greatly reducing blurring.
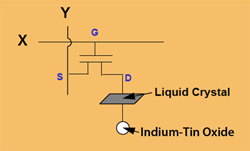 Fig. 12: TFT circuit (Source: Sharp Electronics)
Fig. 12: TFT circuit (Source: Sharp Electronics)
In addition, the second plate (the one farther from the backlight) is coated with a transparent conductive material called indium-tin oxide (ITO). This forms a capacitor with the drain electrodes of the TFTs in the first plate, as shown in Figure 12, increasing the voltage to change the alignment of the molecules, which reduces ghosting.
Of course, in a video image, the field required by each sub-pixel is often different than its neighbors. How can one set of horizontal electrodes and one set of vertical electrodes control each sub-pixel independently? What if two sub-pixels addressed by the same electrode require a different voltage on that electrode?
The answer is surprisingly simple—each frame of the video image is displayed on the screen one row at a time from top to bottom, a process called progressive scanning. This is very similar to progressive raster scanning in an old-style CRT (cathode-ray tube) TV. However, instead of drawing each row from one side of the screen to the other with a beam of electrons as in a CRT, the entire row of sub-pixels in an LCD TV is displayed simultaneously. Then, the next row is activated, and so on until the last row at the bottom of the screen is displayed, after which the entire process is repeated for the next video frame. The horizontal electrode for each row carries a single voltage as it is activated, while the vertical electrodes carry different voltages, providing each sub-pixel in the row with its own, independently controllable electric field.
After each row is displayed, it must remain on the screen with no voltage in the horizontal electrode until the next pass. This requires an additional storage capacitor to be deposited along with each TFT. The capacitor stores just the right amount of electrical energy required for that sub-pixel during that frame, maintaining the liquid-crystal molecules’ orientation until the row is activated again for the next frame.
In Figure 13, you can see a cross-section of the entire sub-pixel structure, along with a circuit diagram with the TFT, storage capacitor (Cs), and capacitor between the TFT and the ITO layer (Clc).
The process is known as sample-and-hold progressive scanning—the electric field for each sub-pixel in a row is created, or “sampled,” and then held until it is updated in the next pass. Interestingly, this is one primary cause of motion blur. (Another, more widely known cause is the response time of the liquid-crystal material.)
Why does sample-and-hold progressive scanning cause motion blur? That will have to wait until the next installment of this article, in which I’ll discuss the various limitations of LCD TVs and how they have been overcome—or not.
Summary: LCD-TVs – Part I
The technology I’ve described in this installment is called TN (twisted-nematic) LCD for obvious reasons. (Confusingly, the liquid-crystal fluid is sometimes called TN as well.) This type of display is inexpensive to manufacture and offers fast response times, so it’s often used in computer monitors and laptop screens. However, it suffers from poor viewing angles, so it’s not used in modern LCD TVs. Still, it is fundamental to understanding how these TVs actually work, which I’ll dig into next time. Until then, happy viewing!
Many thanks to Tom Gattinella and Josh Kairoff of Sharp Electronics for their help with this article.


